Studies have shown that both balloon catheter inductions and Polyhydramnios are each individually associated with a higher risk of umbilical cord prolapse. Umbilical cord prolapse, or UCP, is a catastrophic event that can have a devastating effect on a baby. Because of the increased risk of UCP in balloon catheter inductions combined with the increased risk of UCP with Polyhydramnios, I very strongly believe that balloon catheters should never be used as a method of induction for moms with Polyhydramnios. Before we dive into the rest of the article, here are my top two reasons why:
- one major manufacturer of balloon catheters has stated right on their packaging that they should not be used in patients with Polyhydramnios.
- As the authors of Optimal Management of Umbilical Cord Prolapse stated, “Knowledge of the risk factors of UCP does not significantly decrease its occurrence; however, the anticipation of this problem can lead to improvement of fetal morbidity and mortality. Of note, cord prolapse can occur in pregnancies without obvious risk factors, which renders this complication unpreventable.” (Emphasis added.)
Table of Contents
Medical Disclaimer
The information on this page has not been recognized or verified by any major obstetric organization.
However, it has been gathered from both published, scholarly articles about this topic, and from mothers who have experienced these things first hand. References to published studies are cited where appropriate within the text.
Before making any decision regarding your care, please consult your healthcare provider.
This information has been made available to you so that you can make informed decisions about your care plan because the bottom line is, no one knows you and your body better than you.
What is Umbilical Cord Prolapse?
Umbilical cord prolapse, or UCP, is when the umbilical cord slips into the birth canal either before or alongside your baby’s presenting part. THIS IS A MEDICAL EMERGENCY because it puts the umbilical cord in a position to become compressed between your baby and your bony pelvis, depriving your baby of the oxygen and nutrients it depends on.
A compressed cord causes carbon monoxide to quickly build up in the baby’s bloodstream leading to respiratory acidosis (acid build-up that causes damage to the heart muscle).
Cord compression can also lead to
- brain damage
- fetal hypoxia (oxygen deprivation)
- long term disability, and
- the death of your baby
When umbilical cord prolapse is diagnosed delivery should quickly follow, ideally within 30 minutes. The mortality rate of cord prolapse is also much more favorable when the prolapse happens inside of the hospital (3%) vs. outside (44%).(Sayed)
What Increases the Risk for Cord Prolapse?
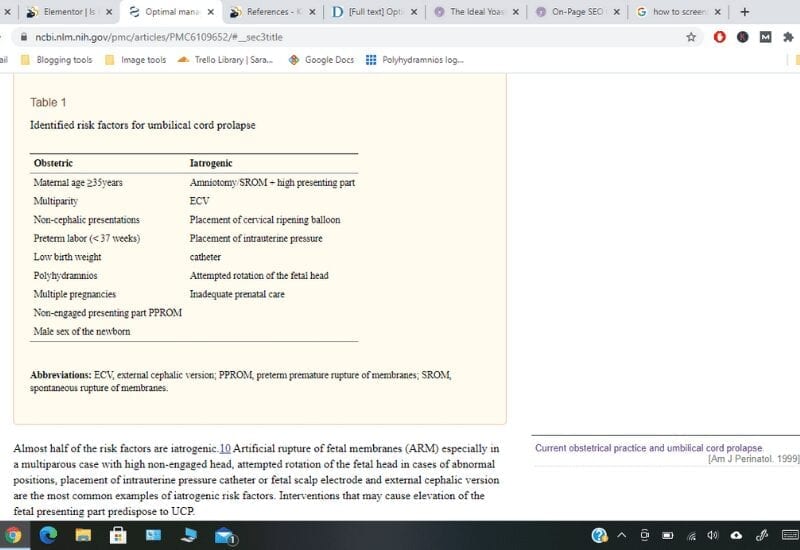
According to an NCBI article on the optimal management of umbilical cord prolapse, there are two categories of risk factors: obstetric (which can not be avoided) and iatrogenic (which can).
Obstetric risk factors include:
- advanced maternal age
- having more than one pregnancy
- fetal malpresentation
- preterm labor
- low birth weight baby
- Polyhydramnios
- being pregnant with multiples
- baby’s head not engaged with PPROM, and
- having a male baby
Iatrogenic risk factors include:
- artificial ROM without baby being engaged
- ECV
- placement of a cervical ripening balloon
- placement of an intrauterine pressure catheter
- manipulation to rotate fetal head, and
- poor prenatal care
Cord Prolapse with Polyhydramnios
As seen above, Polyhydramnios is considered an obstetric (unavoidable) risk factor for cord prolapse. The tricky thing about Polyhydramnios is that the environment it creates also predisposes patients to other obstetric risk factors for cord prolapse including malpresentation, preterm labor, SROM, and non-engaged presenting parts during rupture of membranes (ROM). This is dangerous because the more risk factors you have, the greater your risk.
The following references reveal more about cord prolapse with polyhydramnios:
- Multiple studies of umbilical cord prolapse have identified Polyhydramnios as a significant risk factor for experiencing UCP (Dilbaz, Kaymak, Hasegawa).
- Hasegawa’s nationwide study in Japan determined that of patients who experienced UCP, they were 2.89 times more likely to also have experienced Polyhydramnios.
Cord Prolapse with Balloon Catheter Induction
Remember as you read this section that cervical ripening balloons are considered an iatrogenic risk factor for cord prolapse. Iatrogenic is a term used to describe a medical condition that occurs as a direct result of a medical procedure or the action of a physician.
The following case studies demonstrate the risk of cord prolapse with balloon catheters:
- According to a 1999 study of obstetric practice and umbilical cord prolapse, nearly 1/2 of the cases of UCP were preceded by obstetrical intervention . Of those 41 cases of UCP, 14% used balloon catheter induction (Usta).
- A survey taken in Japan of more than 2 million deliveries over a four year period found that 39% of cases of UCP occurred during the removal of a balloon catheter. The survey concluded, “the risk of umbilical cord prolapse was significantly increased during the use of balloons for cervical ripening (Hasegawa).”
- In our effort to include the most accurate research, we want to include that there was a follow up survey conducted in Japan that concluded, “although the frequency of cervical ripening balloon use significantly declined, the incidence of UCP did not significantly reduce in the last 5 years (Hasewaga).” However, we feel it is important to point out that this survey only included 57% (barely half) of all delivering institutions, and that this is the only study we were able to find with similar results. We also feel that this study proves the point of the quote from the introduction that UCP is an unpreventable complication.
Balloon Catheter Induction with Polyhydramnios
A 2016 study to determine whether balloon catheters were effective for VBAC followed 68 women with a history of prior c-section. The results showed only 1 instance of cord prolapse and determined that for the purpose of vbac, balloon catheter induction “appears a safe option with a good success rate and few maternal and fetal complications.”
However, within this study, there were 12 patients who also had Polyhydramnios, and the ONE instance of UCP happened within this population. That’s 1 in 12, or 8.3%! This mom experienced a SROM that resulted in an emergency c-section only 30 minutes after the insertion of the catheter. Her AFI was 26.3, which is considered mild polyhydramnios (Gonsalves, results, table 2). This study demonstrates a need for further studies specifically looking at the safety of balloon catheters with polyhydramnios.
In addition to this study, consider the following:
The University of South Florida, Boston Medical Center, and Zuckerberg San Francisco General Hospital all list Polyhydramnios as a contraindication for outpatient foley cervical ripening (Outpatient Cervical, Outpatient Foley, Otway). In other words, they only recommend use of a foley catheter under close hospital surveillance.
And perhaps the most compelling piece of evidence is that the only balloon catheter actually approved for use in cervical ripening, the Cook’s Catheter, lists Polyhydramnios as a contraindication for use right in the manufacturer’s user guide (Cook Medical).
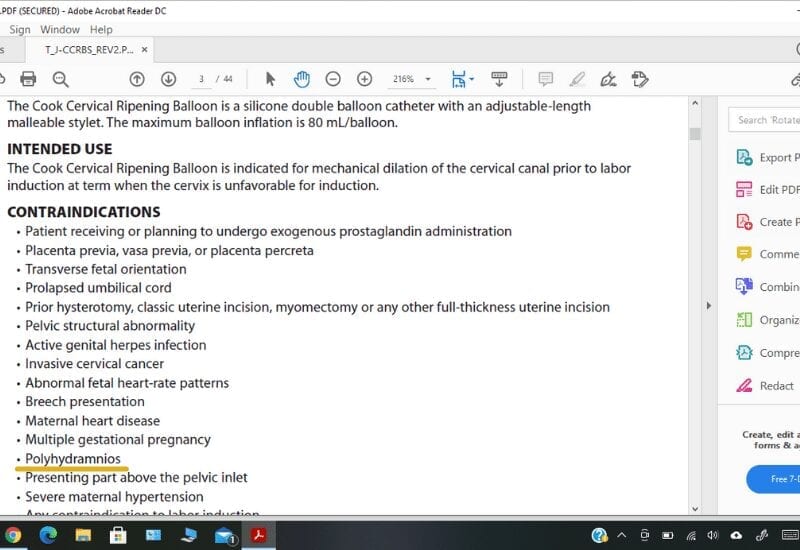
For reference, there are currently two types of balloon catheters available. The foley balloon was not created for and is not licensed for cervical ripening, so it’s use is considered “off label”. The other available catheter is the Cook’s Catheter which has a double balloon and has been licensed for use in cervical ripening.
In addition to these published studies, a quick search through Polyhydramnios support groups online also reveals several more cases of balloon catheter inductions in Poly mamas that resulted in umbilical cord prolapse. We have not included testimonials from these moms in order to respect their privacy.
Takeaway
*Umbilical cord prolapse is a catastrophic event.
*Polyhydramnios is an unavoidable factor that raises the risk for UCP.
*Polyhydramnios also raises the risk of having other UCP factors.
*The more risk factors you have, the higher your risk of having UCP.
*Balloon catheters have been proven to significantly increase the risk of UCP.
*Balloon catheters are an iatrogenic risk factor, meaning it can be avoided.
*Several major hospitals recommend that outpatient foley cervical ripening not be used in moms with Polyhydramnios.
*The manufacturer for the only approved cervical ripening balloon has stated that Polyhydramnios is a contraindication for its use.
Due to the nature of Polyhydramnios, and the fact that adverse outcomes such as UCP are much more common, we feel that the introduction of this particular iatrogenic factor is incredibly risky and not in the best interest of mom or baby. In light of this evidence, we very strongly feel that balloon catheters are not a safe option for the induction of labor with Polyhydramnios.
5 Reasons not to use Balloon Catheter Induction with Polyhydramnios